16.1
Biological Treatments for Mental Disorders
For hundreds of years, people have tried to identify the origins of mental illness, attributing the causes at various times to evil spirits, pressure in the skull, disease, or bad environments. Today, biological explanations and treatments are dominant, partly because of evidence that some disorders have a genetic component or involve a biochemical or neurological abnormality, and partly because physicians and pharmaceutical companies have been aggressively promoting biomedical solutions.

In the years before the advent of antipsychotic medication, patients with severe mental disorders were often put in straitjackets or chained to their beds to keep them from harming themselves or others.
The Question of Drugs
The most commonly used biological treatment is medication that alters the production of or response to neurotransmitters in the brain. Because drugs are so widely advertised and prescribed these days, both for severe disorders such as schizophrenia and for more common problems such as anxiety and depression, consumers need to understand what these drugs are, how they can best be used, and their limitations. Biomedical therapies are discussed in more detail in the video Therapies in Action 1.
Watch
Therapies in Action 1
Drugs Commonly Prescribed for Mental Disorders The main classes of drugs used in the treatment of mental and emotional disorders are:
Antipsychotic drugs, also called neuroleptics—older ones such as Thorazine and Haldol and second-generation ones such as Clozaril, Risperdal, Zyprexa, and Seroquel—are used primarily in the treatment of schizophrenia and other psychoses. However, antipsychotic drugs are increasingly being prescribed “off label” for people with nonpsychotic disorders, such as major depression, bipolar disorder, posttraumatic stress disorder (PTSD), autism, attention-deficit disorder, and dementia.
Most antipsychotic drugs are designed to block or reduce the sensitivity of brain receptors that respond to dopamine; some also block serotonin. Antipsychotic drugs can reduce agitation, delusions, and hallucinations, and they can shorten schizophrenic episodes. But they offer little relief from other symptoms of schizophrenia, such as jumbled thoughts, difficulty concentrating, apathy, emotional flatness, or inability to interact with others. This is why medication by itself is rarely sufficient to help people with schizophrenia manage their symptoms.
Antipsychotics often cause troubling side effects, especially muscle rigidity, hand tremors, and other involuntary muscle movements, which can develop into a neurological disorder called tardive (late-appearing) dyskinesia. In addition, Zyprexa, Risperdal, and other antipsychotics, which manufacturers have been targeting for children and older adults, often carry unacceptable risks for these very groups. The immediate side effect is extreme weight gain—anywhere from 24 to 100 extra pounds a year—which has led to the development of thousands of cases of diabetes; other risks include stroke and death from sudden heart failure (Masand, 2000; Suzuki & Uchida, 2014; Wallace-Wells, 2009).
Although the newer drugs now comprise 90 percent of the market for antipsychotics, a large federally funded study found that they are not significantly safer or more effective than the older, less expensive medications for schizophrenia, the only disorder for which they were originally approved (Lieberman et al., 2005; Swartz et al., 2007). And although antipsychotics are sometimes used to treat impulsive aggressiveness associated with attention-deficit disorder, dementia, and other mental problems, they are ineffective for these disorders. One study followed 86 people, ages 18 to 65, who were given Risperdal, Haldol, or a placebo to treat their aggressive outbursts (Tyrer et al., 2008). The placebo group improved the most. Antipsychotics, which are being given to about one-fifth of combat veterans suffering from PTSD, are also ineffective in reducing symptoms of this disorder (Krystal et al., 2011).

Antipsychotic drugs can help some people with schizophrenia live normal lives. At left, Danny Dunn (seated) poses with her mother. Danny was diagnosed as having schizophrenia and bipolar disorder when she was 17, but medication, therapy, and family support help her function. “I still have challenges and problems,” Danny says, “but life is so much better than it used to be.” The photo on the right shows USC law professor Elyn Saks, who also benefited from medication and therapy, and wrote a memoir of her “journey through madness.” She received a MacArthur Foundation “genius grant” for her contributions to mental health law.
Antidepressant drugs are used primarily in the treatment of depression, anxiety, phobias, and obsessive–compulsive disorder. Monoamine oxidase inhibitors(MAOIs), such as Nardil, elevate the levels of norepinephrine and serotonin in the brain by blocking or inhibiting an enzyme that deactivates these neurotransmitters. Tricyclic antidepressants, such as Elavil and Tofranil, boost norepinephrine and serotonin levels by preventing the normal reabsorption, or “reuptake,” of these substances by the cells that have released them. These older antidepressants are usually more effective for severe depression than the more recent and popular ones, called selective serotonin reuptake inhibitors (SSRIs), such as Prozac, Zoloft, Lexapro, Paxil, and Celexa (Healy, 2012). SSRIs work on the same principle as the tricyclics but specifically target serotonin; Cymbalta and Remeron target both serotonin and norepinephrine. Wellbutrin is chemically unrelated to the other antidepressants but is often prescribed for depression and sometimes as an aid to quit smoking.
All antidepressants tend to produce some unpleasant physical reactions, including dry mouth, headaches, constipation, nausea, restlessness, gastrointestinal problems, weight gain, and, in as many as one-third of all patients, decreased sexual desire and blocked or delayed orgasm (Hollon, Thase, & Markowitz, 2002). The specific side effects may vary with the particular drug. MAOIs interact with certain foods (such as cheese) and they can elevate blood pressure in some individuals to dangerously high levels, so they have to be carefully monitored. Some MAOIs and SSRIs can also produce tardive dyskinesia, although the risk is less than that associated with antipsychotic medications.
Although antidepressants are said to be nonaddictive, all of them, especially the SSRIs, can produce physical dependence, which may feel the same as an addiction to the person taking them. These medications should not be stopped abruptly without supervision by a physician because physical and emotional withdrawal symptoms may occur—including depression and anxiety, which can be mistaken for a relapse, and even mania, which can then cause the sufferer to be misdiagnosed as having bipolar disorder (Kirsch, 2010; Whitaker, 2010).
Antidepressants and most other psychotropic drugs (i.e., those that influence a person’s mental state) now carry strong warnings about the risks of inducing suicide and violence. Of course, some severely depressed people are suicidal, and these drugs can be helpful in alleviating that impulse. But the warnings mean that the clinician and the patient need to be aware that a person on these medications may get worse; if that happens, medication should immediately be reassessed (Healy, 2012).
Anti-anxiety drugs (tranquilizers)—such as Valium, Xanax, Ativan, and Klonopin—increase the activity of the neurotransmitter gamma-aminobutyric acid (GABA). Tranquilizers may temporarily help individuals who are having an acute anxiety attack, but they are not considered the treatment of choice over time. Symptoms often return if the medication is stopped, and a significant percentage of people who take tranquilizers overuse them and develop problems with withdrawal and tolerance (i.e., they need larger and larger doses to get the same effect). Beta blockers, a class of drugs primarily used to manage heart irregularities and hypertension, are sometimes prescribed to relieve acute anxiety—for example, caused by stage fright or athletic competition—which they do by slowing the heart rate and lowering blood pressure. But beta blockers are not approved for anxiety disorders.
A special category of drug, a salt called lithium carbonate, often helps people who suffer from bipolar disorder, although how it produces its effects is unknown. Lithium must be given in exactly the right dose, and bloodstream levels of the drug must be carefully monitored because too little will not help and too much is toxic; in some people, lithium produces short-term side effects (tremors) and long-term problems (kidney damage) (Kemp, 2014). Other drugs commonly prescribed for people with bipolar disorder include Depakote and Tegretol.
For a review of these drugs and their uses, see Review16.1.
Some Cautions about Drug Treatments Without question, drugs have rescued some people from emotional despair and helped countless others live with chronic problems such as schizophrenia, obsessive–compulsive disorder, and panic attacks. They have enabled people suffering from severe depression or mental disturbances to be released from hospitals, to function in the world, and to respond to psychotherapy. Yet many psychiatrists and drug companies are trumpeting the benefits of medication without informing the public of its limitations.
Most people are unaware of how a publication bias—the tendency for journals to publish positive findings but not negative or ambiguous ones—affects what we know. Independent researchers were able to obtain unpublished data submitted to the U.S. Food and Drug Administration (FDA) on 12 popular antidepressants, and you can see the surprising results they uncovered in Figure16.1. Of the 38 studies that reported positive results, 37 were later published. Of the 36 studies with negative or mixed results, only 14 were published (Turner et al., 2008). Even more worrisome for the prospects of impartial research, the majority of researchers who are studying the effectiveness of medication have financial ties to the pharmaceutical industry, in the form of lucrative consulting fees, funding for their clinical trials, stock investments, and patents. Studies that are independently funded often do not get the positive results that industry-funded drug trials do (Angell, 2004; Healy, 2002; Krimsky, 2003). In this section, therefore, we want to give you an idea of what you are not hearing from the drug companies.
Figure 16.1
Drugs and Publication Bias
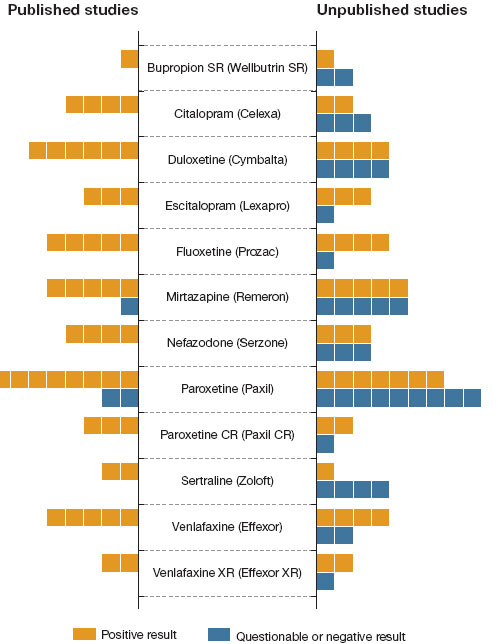
To get FDA approval for a new medication, a pharmaceutical company must present evidence of the drug’s effectiveness. On the bars in this figure, each box represents one study. On the left side, you can see that most of the published studies supported the effectiveness of 12 antidepressants. But when independent researchers got hold of all of the data submitted to the FDA, they found that many unpublished studies had questionable or negative results (right). (Based on Turner et al., 2008.)
The placebo effect. New drugs often promise quick and effective cures. But the placebo effect ensures that many people will respond positively to a new drug just because of the enthusiasm surrounding it and because of their own expectations that the drug will make them feel better. After a while, when placebo effects decline, many drugs turn out to be neither as effective as promised nor as widely applicable. This has happened repeatedly with each new generation of tranquilizer and each new “miracle” antipsychotic drug and antidepressant (Healy, 2012; Moncrieff, 2001, 2013).
In fact, considerable evidence shows that much of the effectiveness of antidepressants, especially for people who are only mildly depressed, is due to a placebo effect (Khan et al., 2003; Kirsch, 2010). Overall, only about half of all depressed patients respond positively to any given antidepressant medication, and of those, fewer than half are actually responding to the specific biological effects of the drug (Hollon, Thase, & Markowitz, 2002). A meta-analysis of more than 5,000 patients in 47 clinical trials revealed that the placebo effect was “exceptionally large,” accounting for more than 80 percent of the alleviation of symptoms. The drugs were most effective for patients with severe depression (Kirsch et al., 2008). The psychological expectation of improvement by taking a placebo actually produces some of the same brain changes that medication does (Benedetti et al., 2005).
Another line of evidence supports the argument that, for most people, the benefits of antidepressants are unrelated to their chemical action: Higher doses of the drugs do not produce greater reductions in depressive symptoms, only more side effects (Kirsch, 2010). But when the drug isn’t working, its manufacturer generally advises the prescribing physician to increase the dose.
High relapse and dropout rates. A person may have short-term success with antipsychotic or antidepressant drugs. However, in part because of these drugs’ unpleasant side effects, anywhere from one-half to two-thirds of people stop taking them. When they do, they are likely to relapse, especially if they have not learned how to cope with their disorders (Hollon et al., 2002).
One reason some patients may stop taking a drug is that they have been given the wrong dose. The same dose may be metabolized differently in men and women, old people and young people, and Asians and African Americans or Anglos (Lin, Poland, & Chien, 1990; Strickland et al., 1995). Groups may differ in the dosages they can tolerate because of variations in metabolic rates, amount of body fat, the number or type of drug receptors in the brain, or cultural differences in smoking and diet.
Disregard for effective, possibly better nonmedical treatments. The popularity of drugs has been fueled by pressure from managed-care organizations, which prefer to pay for one patient visit for a prescription rather than 10 visits for psychotherapy, and by drug company marketing and advertising. In 1997, the FDA permitted pharmaceutical companies to advertise directly to the public, a practice still forbidden in most of the rest of the world; sales of new drugs skyrocketed as consumers began to request them. Because ads promise such wonderful results, medication often seems the best way to deal with an emotional or behavioral problem; yet nonmedical treatments may work just as well or better. Consider this study of more than 168,000 children who had been referred to a behavioral-care facility to be treated for attention-deficit disorder. More than 60 percent of the boys and 23 percent of the girls were on Ritalin or another drug. But after six sessions of behavior therapy for the children and 10 sessions for the parent, only 11 percent of the boys and 2 percent of the girls remained on medication (Cummings & Wiggins, 2001; see also March, 2011).
Unknown risks over time and drug interactions. The effects of taking antidepressants indefinitely are still unknown, especially for vulnerable groups such as children, pregnant women, and older adults—or for the generation of young adults who have been taking them since childhood or adolescence, when the brain is still developing. After British drug authorities reported that nine unpublished studies of Paxil found that it tripled the risk of suicidal thoughts and suicide attempts in adolescents who were taking the drug compared to those given a placebo, the FDA now warns against prescribing SSRIs to anyone under age 18.
The reason we don’t know about long-term effects until a drug has been on the market for years is that new drugs are initially tested clinically on just a few hundred people for just a few weeks or months, even when the drug is one that a person might take indefinitely (Angell, 2004). (The cost of bringing most new drugs to market is very high, and manufacturers feel they cannot afford to wait years to determine whether there might be long-term hazards.) Nonetheless, many psychiatrists, understandably frustrated by the failure of existing antipsychotics and antidepressants to help all of their patients, are prescribing “cocktails” of medications—this one for anxiety, plus this one for depression, plus another to manage the side effects. They report anecdotal success in some cases, but as yet there has been virtually no research on the benefits and risks of these combination approaches.
Untested off-label uses. Most consumers do not realize that after the FDA approves a drug, doctors are permitted to prescribe it for other conditions and to populations other than those on which it was originally tested. As already noted, antipsychotics such as Risperdal are being used for nonpsychotic disorders. Likewise, antidepressants are being marketed for “social phobias”; Prozac, when its patent expired, was renamed Sarafem and marketed to women for “premenstrual dysphoric disorder”; and Ritalin, widely given to school-aged children, is being prescribed for 2- and 3-year-olds.
In coming years, you will be hearing about many “promising medications” for such common psychological problems as memory loss, eating disorders, smoking, and alcoholism. Every large pharmaceutical company is working on one or more of these. But we hope you will resist the impulse to jump on any new drug bandwagon. Critical thinkers must weigh the benefits and limitations of any medication for psychological problems; wait for the data on safety and effectiveness; and resist the temptation to oversimplify.
Direct Brain Intervention
For most of human history, a person suffering from mental illness often got a rather extreme form of “help.” A well-meaning tribal healer or, in later centuries, a doctor, would try to release the “psychic pressures” believed to be causing the symptoms by drilling holes in the victim’s skull. It didn’t work! To learn more about the history of therapeutic “cures,” watch the video Assessing Treatment Effectiveness.
Watch
Assessing Treatment Effectiveness
The most famous modern effort to cure mental illness through psychosurgery—intervening directly in the brain—was invented in 1935, when a Portuguese neurologist, António Egas Moniz, drilled two holes into the skull of a mental patient and used an instrument to crush nerve fibers running from the prefrontal lobes to other areas. (Later, some doctors just used an ice pick.) This operation, called a prefrontal lobotomy, was supposed to reduce the patient’s emotional symptoms without impairing intellectual ability. The procedure—which, incredibly, was never assessed or validated scientifically—was performed on more than 40,000 people in the United States, including President John F. Kennedy’s sister Rosemary. Tragically, lobotomies left many patients apathetic, withdrawn, and unable to care for themselves (Dully, 2008; Raz, 2013; Valenstein, 1986). Yet, Moniz won a Nobel Prize for his work.
In contrast to surgical intervention, some psychiatrists now attempt to alter brain function by stimulating the brain externally. The oldest method is electroconvulsive therapy (ECT), or “shock therapy,” which is used for the treatment of severe depression, although no one knows how or why it works. An electrode is placed on one side of the head and a brief current is turned on. The current triggers a seizure that typically lasts 1 minute, causing the body to convulse. In the past, there were many horror stories about the misuse of ECT and its dire effects on memory. Today, however, patients are given muscle relaxants and anesthesia, so they sleep through the procedure and their convulsions are minimized. The World Psychiatric Association and the FDA have endorsed ECT as safe and effective, especially for people with episodes of crippling depression and suicidal impulses and for those who have not responded to other treatments (Shorter & Healy, 2008). About 100,000 Americans receive ECT every year. However, the mood-improving effect of ECT is usually short-lived, and the depression almost always returns within a few weeks or months (Hollon, Thase, & Markowitz, 2002; U.S. Food and Drug Administration, 2011). And ECT is ineffective with other disorders, such as schizophrenia or alcoholism, though it is occasionally misused for these conditions.
Direct Brain Intervention in the Treatment of Disorders
Neuroscientists are investigating other ways of electrically stimulating the brains of severely depressed individuals (Nieudworp et al., 2015; Nitsche et al., 2009). One is transcranial magnetic stimulation(TMS), which involves the use of a pulsing magnetic coil held to a person’s skull over the left prefrontal cortex, an area of the brain that is less active in people with depression. As with ECT, the benefits of TMS, when they occur, are short-lived, but it can be effective with some patients and has fewer side effects than ECT (George & Post, 2011; Prasser et al., 2015).
In recent years, psychosurgery has returned, with neurologists experimenting with procedures on parts of the brain thought to be involved in various disorders that have not responded to any other treatments (Brunoni et al., 2011). One approach is deep brain stimulation(DBS), which originally was approved for patients with Parkinson’s disease and epilepsy; now it is being used for a variety of mental disorders, such as obsessive–compulsive disorder, though again no one knows how or why it might be helpful (Sharma et al., 2015). DBS requires surgery to implant electrodes into the brain and to embed a small box, similar to a pacemaker, under the collarbone. But it also is experimental and risky, and claims of its success are based only on patients’ self-reports, so the powerful placebo effect of surgery cannot be ruled out (Lozano et al., 2008). The company that developed this expensive device has hired psychiatrists to lobby for it and mounted a vigorous marketing campaign to get it approved, but critics are concerned that the method has not been adequately tested, its effectiveness and side effects are unknown, and it promises more than it delivers (Barglow, 2008; Fins et al., 2011).